Natural Der p 2 Molecular Reference Std. (MRS-NDP2-1)
The Natural Der p 2 MRS is intended to serve as reference standard to determine the Der p 2 content of allergen preparations from house dust mite (Dermatophagoides pteronyssinus) by immunoassay.
Allergen: Natural Der p 2 (Dermatophagoides pteronyssinus allergen 2)
Source: D. pteronyssinus culture
Mol. Wt: 14 kD
Purification: From spent mite culture by multi-step affinity chromatography.
Composition: 10µg natural Der p 2, determined by Amino Acid Analysis, freeze dried in sealed glass vial.
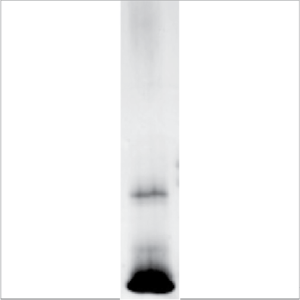
SDS-PAGE: See inset. Silver-stained SDS-PAGE under non-reducing conditions shows a single band at 14kD.
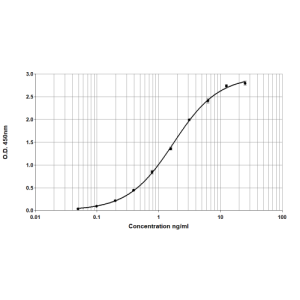
ELISA: Immunoreactive in Der p 2 specific ELISA. No trace contamination with Der p 1 was detected by ELISA.
Purity: >98% purity by in-solution LC-MS/MS after tryptic digest.
Isoforms: Analyzed by LC-MS/MS. Main Isoforms: Der p 2.0110 (43%) and Der p 2.0101 (37%). Other Isoforms present (20%): Der p 2.0103, Der p 2.0104, Der p 2.0114.
Formulation: Preservative-free and carrier-free in phosphate buffered saline, pH 7.4. Filtered through 0.22µ filter.
Storage: Store at -20ºC
Product Resources: Natural Der p 2 Molecular Reference Standard Certificate of Analysis
Allergens are provided for research and commercial use in vitro: not for human in vivo or therapeutic use.
Reconstitution:
- Allow vial to reach room temperature before use
- Tap vial gently to collect all material at the bottom
- Using a sterile syringe reconstitute the MRS to desired concentration by injecting a suitable volume of a buffer of choice (e.g. PBS, pH 7.4 or 1% BSA/50% glycerol/PBS, pH 7.4).
- Mix by gently swirling the vial until content is completely dissolved.
- Adding 1ml of buffer will result in a Der p 2 concentration of 10,000ng/ml.
References:
- Smith AM et al. The molecular basis of antigenic cross-reactivity between the group 2 mite allergens. J Allergy Clin Immunol 2001;107:977-84.
- Derewenda U et al.The crystal structure of a major dust mite allergen Der p 2, and its biological implications. J Mol Biol. 2002;318:189-97.
- Trompette A et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature. 2009;457:585-8.
- Chapman MD et al. The European Union CREATE project: a model for international standardization of allergy diagnostics and vaccines. J Allergy Clin Immunol. 2008;122:882-889.
- van Ree R et al. The CREATE project: development of certified reference materials for allergenic products and validation of methods for their quantification. Allergy. 2008;63(3):310-26.
- Kaul S et al. Regulatory environment for allergen-specific immunotherapy. Allergy 2011;66:753-64.
- Chapman MD and Briza P. Molecular approaches to allergen standardization. Curr Allergy Asthma Rep. 2012;12:478-84.
- Chapman MD et al. Technological Innovations for High-Throughput Approaches to In Vitro Allergy Diagnosis. Curr Allergy Asthma Rep. 2015;15:36